Which Element Has Similar Properties to Lithium?
In the rapidly evolving world of residential energy storage and industrial power, understanding the fundamental chemistry of your battery is key to long-term ROI. The question "which element has similar properties to lithium?" isn't just for scientists; it's the foundation for choosing between established performance and experimental alternatives. By exploring lithium location on the periodic table, we can decode why certain systems deliver superior power density and ease of use.
Which element has similar properties to lithium in terms of atomic structure?
To identify what elements are similar to lithium, we must look at Group 1, the Alkali Metals. Elements in this family, such as Sodium (Na) and Potassium (K), share the same "Chemical DNA"—a single valence electron. This shared trait makes them highly reactive and capable of forming +1 cations, which is the basic requirement for ion-transfer batteries.
However, a more sophisticated answer to which element is chemically similar to lithium lies in the "Diagonal Relationship." Lithium shares a unique parity with Magnesium (Mg). Because their ionic radii are remarkably similar ($0.76\text{ \AA}$ for Li vs. $0.72\text{ \AA}$ for Mg), they exhibit comparable reactivity and solubility patterns. Yet, while these siblings look similar on paper, their physical performance in a real-world battery environment tells a different story.
Rubidium (Rb):Located below lithium in the alkali metal family, rubidium shares many similarities with lithium. They both have strong reactivity, readily form +1 ions, and impart a unique red or violet color to their respective salts' flames.
Beryllium (Be): Although beryllium is an alkaline earth metal, it shares some similarities with lithium despite being located below it on the periodic table. They both have two valence electrons and form +2 cations. Their oxides and hydroxides also have relatively high melting points.
Magnesium (Mg): Like beryllium, magnesium is an alkaline earth metal that also shares some similarities with lithium. They both have similar electronegativity and reactivity, but magnesium forms +2 ions when reacting.
Calcium (Ca): Like magnesium, calcium also forms +2 ions. Compared to most other elements, lithium and calcium have relatively low first and second ionization energies.
In summary, the elements most similar to lithium in terms of reactivity and properties are the alkali metals sodium, potassium, and rubidium. Beryllium and magnesium also exhibit some similarities.

Why are sodium and lithium similar elements, and how do they differ in performance?
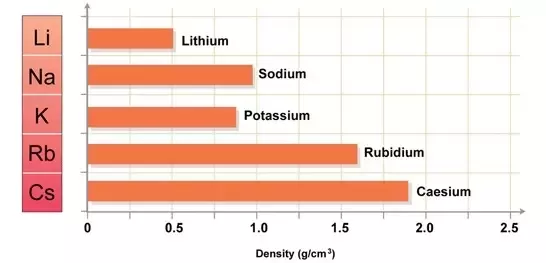
Understanding why are sodium and lithium similar elements requires looking at their physical profiles. Both are ultra-lightweight metals that can float on water (Lithium at $0.534\text{ g/cm}^3$ and Sodium at $0.971\text{ g/cm}^3$). Despite this similarity, the atomic radius of Sodium ($102\text{ pm}$) is significantly larger than Lithium’s ($76\text{ pm}$).
This size difference is not just a detail; it is a performance barrier. Peer-reviewed research in the Journal of Power Sources confirms that Sodium's larger ionic size directly inhibits its migration speed through the battery’s cathode lattice. This "atomic traffic jam" results in slower charging and lower power delivery. This is why high-performance, independent units like the Batterlution PowerGo Series continue to rely on the refined efficiency of lithium. By harnessing the fastest-moving ions, the PowerGo achieves a level of responsiveness that allows it to function as a simple, standalone device—connected via a standard Schuko plug directly into your AC socket without the need for complex, slow-reacting hardware.
Why are lithium-ion batteries more popular than sodium or potassium alternatives?
If you're wondering why are lithium-ion batteries more popular, the answer is "Electrode Potential." Lithium has the lowest standard potential of any metal ($-3.04\text{V}$), which creates a much higher energy density compared to its similar elements.
This high energy density is what makes true "Plug & Play" storage possible. Because lithium-ion can pack more power into a smaller space, the Batterlution PowerGo can remain a compact, independent unit. Unlike traditional bulky installations, it features a simple Schuko plug connection to AC power outlets, making it the most retrofit-friendly solution for homes with existing PV systems. By optimizing energy usage through a certified, robust BMS, this system allows homeowners to slash their energy bills by up to €1,200/year using the most energy-dense chemistry available today.
- High Ionization Energy Characteristics: Lithium-ion batteries have significantly higher energy density than other battery types, meaning they can store more energy in a smaller and lighter package. This makes them ideal for applications that demand high-performance batteries, such as mobile devices and electric vehicles.
- Long cycle life: Lithium-ion batteries also have a very long cycle life, meaning they can be used for many years without needing to be replaced. This can reduce overall costs and improve reliability.
- Zero maintenance: Lithium-ion batteries do not require regular maintenance, such as charging or discharging. This can save time and effort.
- Versatility: Lithium-ion batteries can be used in a wide variety of applications, from small electronic devices to large power grids. This makes them a very versatile battery technology.
Where is lithium in the periodic table, and how does its location impact recycling?
The lithium location on the periodic table—at the very top of the reactive metals—makes it a valuable resource that is highly suitable for circular recovery. So, how are lithium-ion batteries recycled, and what happens after? The process involves discharging the cells, shredding them into "black mass," and using hydrometallurgy to extract pure lithium carbonate.
This recycled lithium is then fed back into the production of new, high-performance batteries. As future development trends lean toward solid-state technology, the industry is finding ways to make lithium’s reactive properties even safer by replacing liquid electrolytes with solid ones. This commitment to safety and sustainability is mirrored in the Batterlution philosophy, where every PowerGo unit is built with all-in-one safety certifications to ensure that the raw power of Group 1 elements is handled with industrial-grade precision.
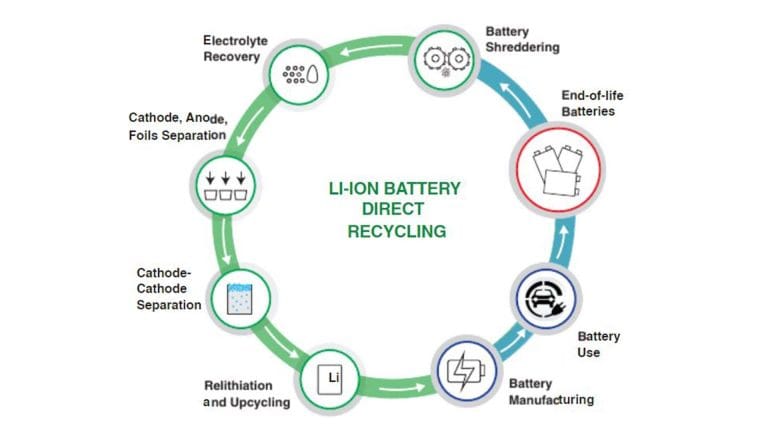
The steps are as follows:
- Disassembly: Batteries are carefully disassembled to recover individual components, such as cathodes, anodes, electrolyte, and casing.
- Shredding: Disassembled batteries are shredded to further separate the components.
- Valuable battery metals: Valuable metals like lithium, cobalt, and nickel are recovered through valuable battery metals of the battery components.
The recovered materials can be used for various purposes:
- Metal recycling: Recovered metals can be reused in battery production or other industries, reducing the need for raw material extraction.
The steps are as follows:
- Disassembly: Batteries are carefully disassembled to recover individual components, such as cathodes, anodes, electrolyte, and casing.
- Shredding: Disassembled batteries are shredded to further separate the components.
- Valuable battery metals: Valuable metals like lithium, cobalt, and nickel are recovered through valuable battery metals of the battery components.
The recovered materials can be used for various purposes:
- Metal recycling: Recovered metals can be reused in battery production or other industries, reducing the need for raw material extraction.
Which element is chemically similar to lithium for the future of energy?
While we continue to search for which element has similar properties to lithium to lower costs, lithium remains the king of efficiency and simplicity. The Batterlution PowerGo Series represents the pinnacle of this chemistry, offering an independent, Schuko-ready solution that works seamlessly with your existing energy setup. It’s not just about finding an element that is "similar"; it's about utilizing the one that works best for your home.

Batterlution PowerGo Series
Plug & Play Solar Battery Storage – Smarter, Simpler, Scalable
• Boost Self-Consumption – Store excess solar energy and slash your bills by up to €1,200/year.
• Retrofit-Friendly – Works seamlessly with existing PV systems (no system overhaul needed).
• Dynamic Power Price Ready – Optimize charging during low-cost hours for extra savings.
• Ultra-Fast Installation – True plug-in design; no electrician required (certified & all-in-one safety).
Contact us
Sodium (Na) is the closest chemical sibling. However, its larger atomic size makes it heavier and less efficient. For an independent unit like the Batterlution PowerGo Series, lithium remains the only choice that balances high energy density with a compact design.
It comes down to efficiency and space. Lithium’s high ionization energy allows for a much smaller footprint. This enables the PowerGo to function as a standalone device with a simple Schuko plug connection to AC power outlets, avoiding the bulky hardware required by similar elements.
Yes, lithium is reactive, but professional engineering makes it safe. The Batterlution PowerGo uses all-in-one safety certified LFP chemistry and a robust BMS to tame lithium’s properties, making it safer for indoor use than DIY setups using other alkali metals.
Lithium sits at the top of Group 1, giving it the fastest ion mobility. This allows the PowerGo to react instantly to dynamic power prices, charging and discharging faster than its “similar element” rivals to save you up to €1,200/year.









